Sanofi: Streamlining Clinical Study Training
Across the Portfolio
30%
projected savings
3
systems eliminated
10,000
users across 43 countries
Study training is a significant yet often hidden cost in clinical trials. For an average Phase 2 study with 40 sites, study training costs can exceed $200,000 – assuming that everyone completes training once and correctly. With retraining, that figure can jump to $350,000.
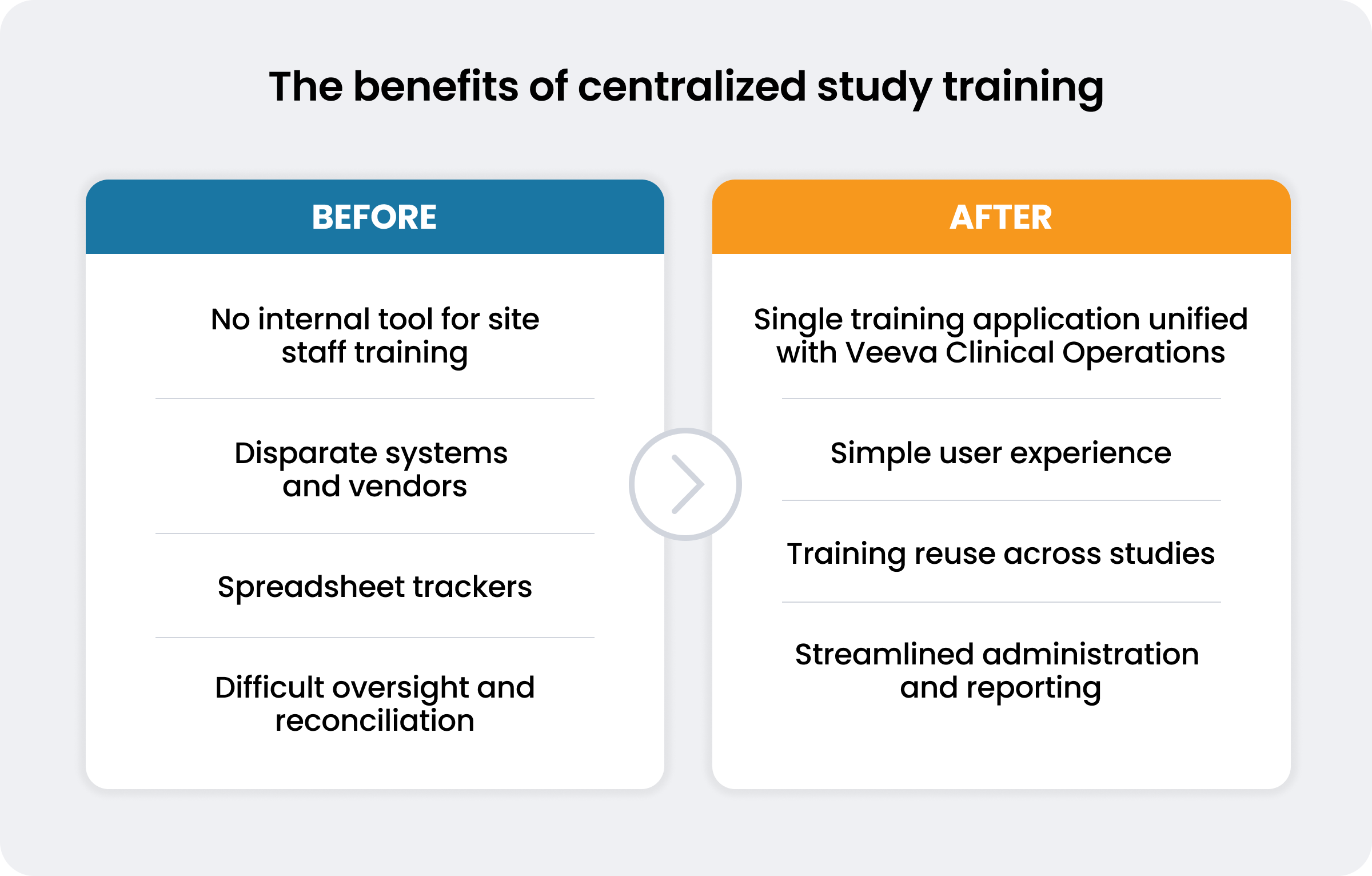
Delphine Maillard, head of clinical learning and development at Sanofi, knew these challenges well. As a top 10 biopharma with over 80,000 employees, Sanofi lacked an internal system to train site staff. Instead, Maillard’s team relied on multiple, disparate systems provided by service providers.
“When you don’t have a centralized system, it’s not possible to reconcile all the training information. Ultimately, that is what brought us to Veeva Study Training,” explains Maillard. Since consolidating three previous systems into a single platform, the company is on track to reduce study training costs by an estimated 30%.
Saving time with automated study training workflows
Maillard points to several Veeva Study Training capabilities that help Sanofi save time and improve oversight:
- Training matrix: Team members can automatically assign training based on user roles and responsibilities. This is especially helpful when new team members join a study, ensuring they receive the necessary training without delay. “You can create a draft of the training matrix, then study managers approve roles and training assignments. Once validated, the system automatically sends notifications to learners,” says Maillard.
- Crosslink with eTMF: When a study manager uploads a document in Veeva eTMF, they can also send it to Veeva Study Training. The system automatically creates the training environment, assigns it through the training matrix, and notifies users. “It’s very quick and easy,” Maillard notes.
- Dynamic dashboards and reports: Real-time dashboards provide visibility into training status, metrics, and activities across studies.

A quick-win to kick off Sanofi’s clinical transformation
For Sanofi, implementing Veeva Study Training was part of a broader initiative to reshape clinical operations. Five years ago, the company began implementing the Veeva Clinical Operations Platform, starting with Veeva eTMF followed by Veeva Study Training.
“We needed a quick win to kick off our multi-year transformation – and Veeva Study Training was just that,” says Maillard.
Sanofi deployed the system to internal users in nine months and went live with site staff just two months later. More than 10,000 users based in 43 countries – including 8,000 site staff members – have access to the system. The company has already completed over 128,000 total training assignments.
“It makes life much easier for site staff. They use one Veeva login to access all Sanofi studies and receive automated notifications when training is required. The system also tracks training that repeats across studies, so site staff don’t have to complete the same modules multiple times.”
Lessons learned from Sanofi’s implementation
Sanofi partnered with Veeva Business Consulting to implement Study Training in phases. For companies considering a new study training system, Maillard recommends avoiding customization wherever possible to support easier integration with existing processes.
She also emphasizes the value of a dedicated planning phase. “We took time to define our project scope, implementation strategy, and operating model,” says Maillard. “It was also an opportunity to review and harmonize our study training processes.
Piloting the ‘study training manager’ role
Like many biopharmas, Sanofi’s study teams were previously responsible for managing study training. During implementation, the company piloted a new role: the study training manager (STM).
STMs define training strategies, manage rollout of the training matrix, and provide first-line support to users. They also ensure consistency and quality across studies. As a result, global study managers can focus on their core responsibilities.
Next steps on Sanofi’s journey toward unified clinical
With study training standardized, Maillard’s team is expanding its STM program and plans to implement Veeva CTMS, Study Startup, Site Connect, and Payments over the next few years. “This journey toward centralized, compliant study training has delivered significant benefits to Sanofi, our sites, and ultimately to our patients,” says Maillard. “By starting with Study Training, we’ve established a solid foundation for future transformation initiatives.”
Learn how another top 10 biopharma streamlined study training and enhanced oversight.
More Customer Stories
Explore and learn more
Read Customer Story
Site Training Soars at Bayer with Veeva Study Training

Watch Video
Novotech: Eliminating Silos and Driving Efficiencies with Veeva Study Training

Watch Video
Boehringer Ingelheim: Driving Efficiency and Collaboration with a Unified Clinical Operations Platform

