Veeva LIMS for Virtual Biopharma
Modernize Quality Control with a Digital-First Solution
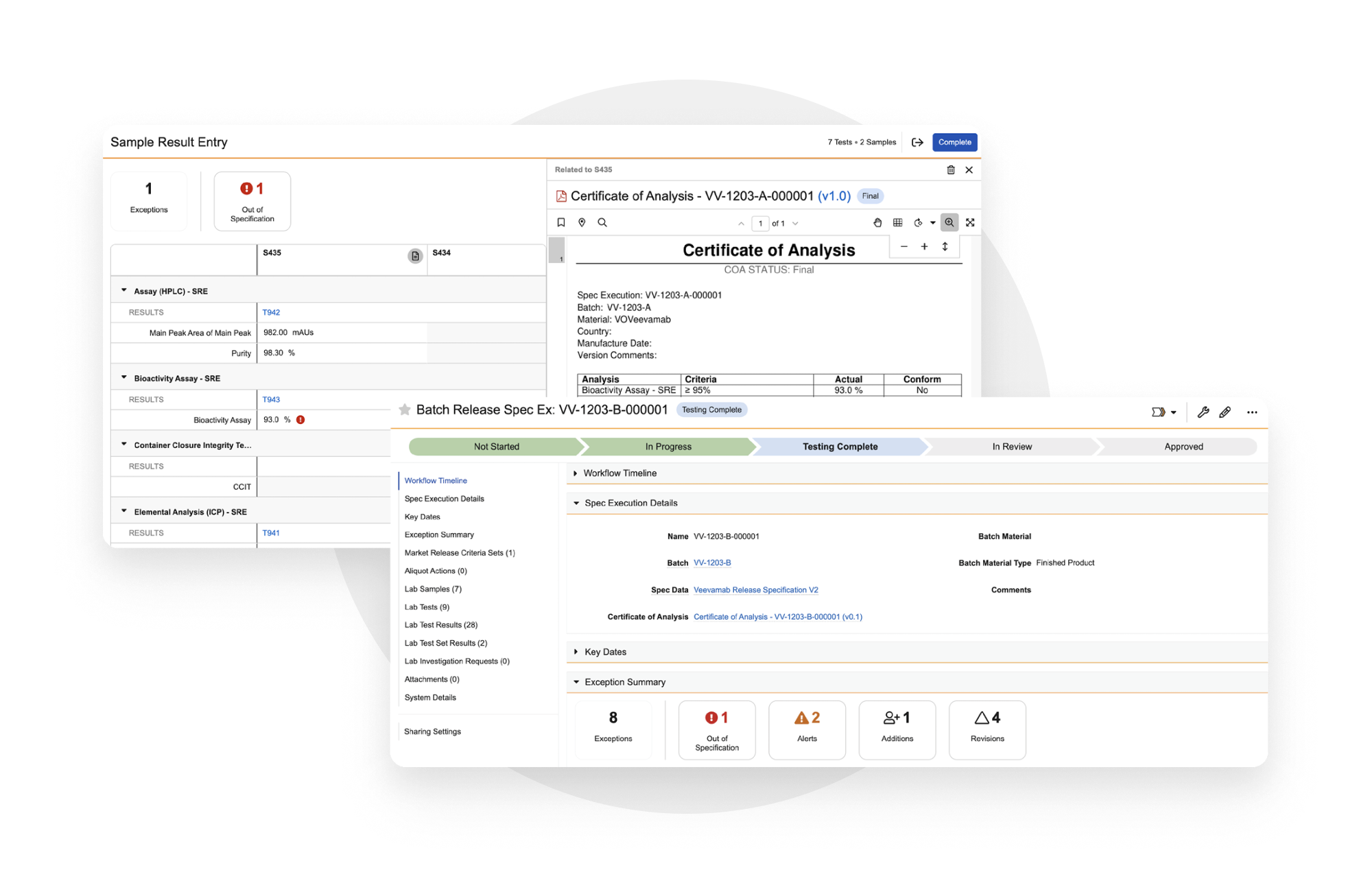
Veeva LIMS is a comprehensive cloud application that unifies QC data management across biopharmas and their contract partners. It enables Quality Control to optimize stability study management, external QC results entry, trend analysis, CoA and Stability report generation, and publication of QC outcomes to Veeva Batch Release to accelerate the release of products.
Alongside other Quality Cloud applications, including QMS, QualityDocs, and Training, LIMS delivers the efficiency, GMP compliance, and scalability companies need for product development and commercial launch.
Announced 2021 Status Early Customers 11-50
Overview
Modernize Quality Control with a Digital-First Solution
Veeva LIMS is a comprehensive cloud application that unifies QC data management across biopharmas and their contract partners. It enables Quality Control to optimize stability study management, external QC results entry, trend analysis, CoA and Stability report generation, and publication of QC outcomes to Veeva Batch Release to accelerate the release of products.
Alongside other Quality Cloud applications, including QMS, QualityDocs, and Training, LIMS delivers the efficiency, GMP compliance, and scalability companies need for product development and commercial launch.
Why Veeva LIMS
Modern, unified QC data management
Resources
Explore and Learn
Read Features Brief
Veeva LIMS for Virtual Biopharma Feature Brief

Read Blog
Stability Studies and Trending

Read Article
Advice from a Growing Biotech on Launching a New LIMS Within a Virtual Organization

Read White Paper
Building the Business Case for LIMS

Read Infographic
A Checklist for LIMS: What Should Your Business Case Cover?

Watch Video
Why Veeva LIMS

